 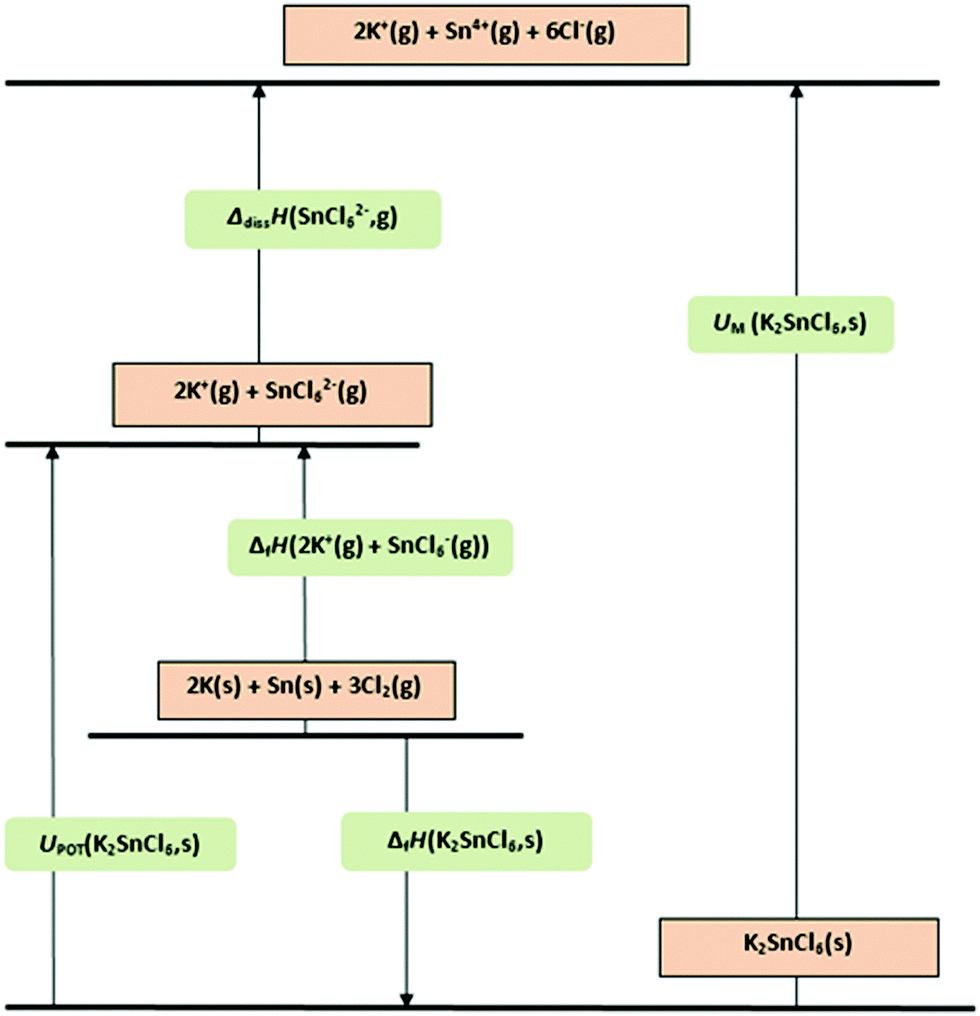
Ionic bonding is an electrostatic attraction between oppositely charged ions. Lattice Enthalpy Bond enthalpies provide a measure of the strength of covalent bonds. Explain, in qualitative terms, the effect of ionic charge and of ionic radius on the numerical magnitude of a lattice energy. 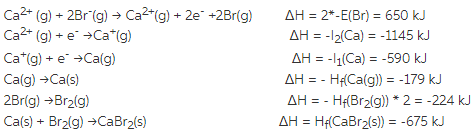
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
